/periodic-table-165930186-590f2d703df78c92832fe141.jpg)
Similarly, as we proceed across the row, the increasing nuclear charge is not effectively neutralized by the electrons being added to the 2 s and 2 p orbitals. In a neutral atom, the number of electrons equals the number of protons. An orbital often is depicted as a three-dimensional region within which there is a 95 percent probability of. The chemistry of each element is determined by its number of protons and electrons. Consequently, beryllium is significantly smaller than lithium. orbital, in chemistry and physics, a mathematical expression, called a wave function, that describes properties characteristic of no more than two electrons in the vicinity of an atomic nucleus or of a system of nuclei as in a molecule. In a group, the chemical elements have atoms with identical valence electron counts and identical valence vacancy counts. This means that the effective nuclear charge experienced by the 2 s electrons in beryllium is between +1 and +2 (the calculated value is +1.66). Group, in chemistry, a column in the periodic table of the chemical elements. Scientists use the periodic table to quickly refer to information about an element, like atomic mass. This entry was posted on Jby Anne Helmenstine (updated on February 1, 2023) A periodic table group is a column, while a periodic table period is a row. There are many observable patterns in elements’ physical and chemical properties as we move across a period or down in a group in the Periodic Table. The periodic table of chemical elements, often called the periodic table, organizes all discovered chemical elements in rows (called periods) and columns (called groups) according to increasing atomic number. Home » Science Notes Posts » Chemistry » Periodic Table » Periodic Table Groups and Periods. These trends are broadly divided into two categories, i.e., physical properties and chemical properties. (More detailed calculations give a value of Z eff = +1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. The elements of the Periodic Table show various trends. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately +1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. Not all elements form stable negative ions in which case the electron affinity is zero or even positive. So the more negative the electron affinity the more favourable the electron addition process is. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. The electron affinity is the potential energy change of the atom when an electron is added to a neutral gaseous atom to form a negative ion. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from +3 to +10. By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior. Element symbols also can refer to alchemy symbols for the elements or to the symbols used to describe. When a symbol consists of two letters, the first letter is always capitalized, while the second letter is lowercase. 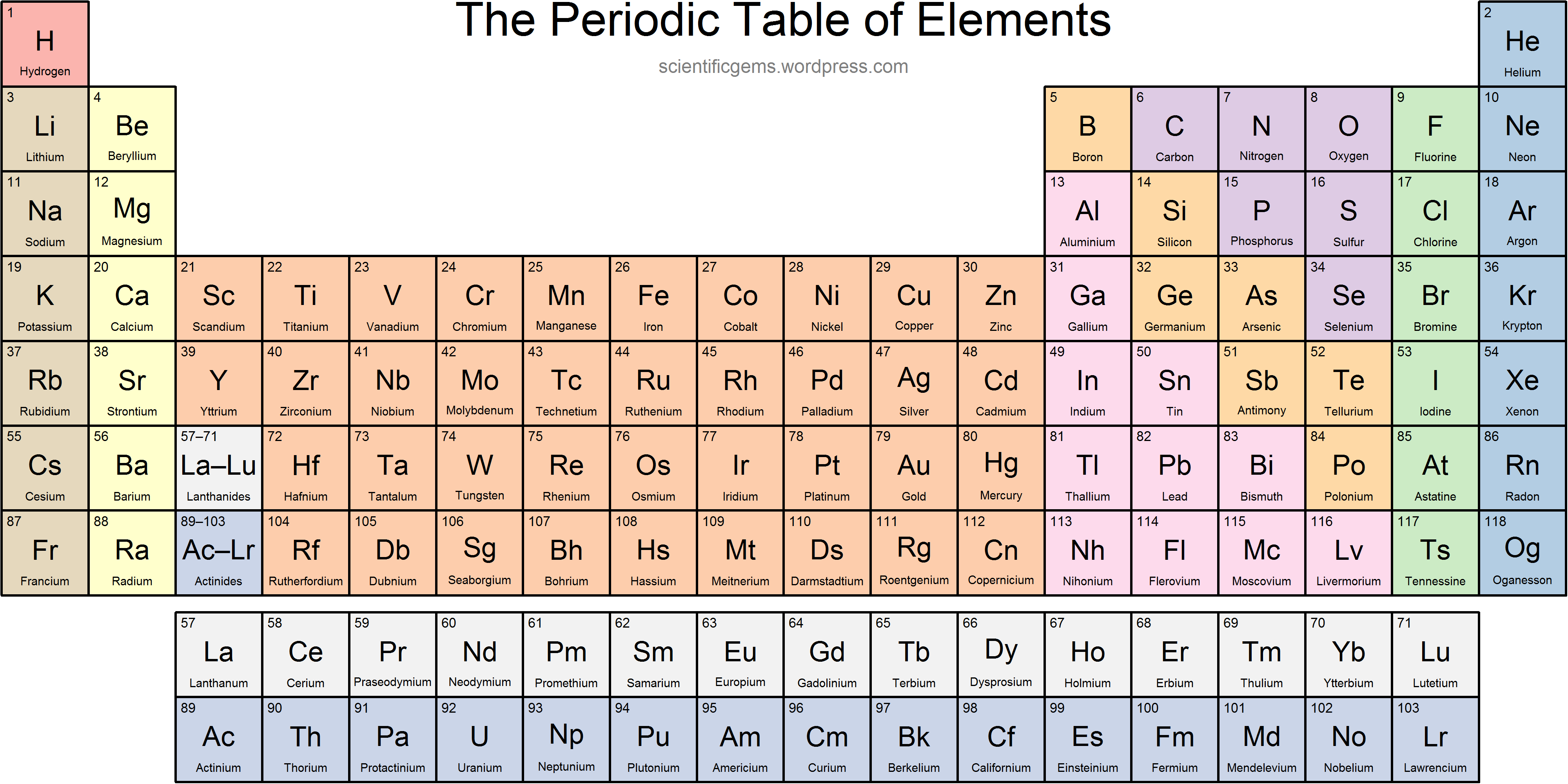
An element symbol is a one- or two-letter abbreviation for a chemical element name. The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. Key Takeaways: Element Symbol Definition. The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. This first video is of bromine reacting with aluminum.\( \newcommand\)) experienced by electrons in the outermost orbitals of the elements. Watch the following two video experiments of \(p\) block elements: They were given the name halogen, from the Greek roots hal - (salt) and - gen (to produce), because they all. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). \) (Credit: Courtesy of NASA Source: (opens in new window) License: Public Domain) halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
